Engineering Decisions Behind a Real Plant System
Chemical engineers convert process requirements into industrial systems that can operate safely, consistently, and under control. In practice, this means defining process conditions, selecting equipment, setting operating limits, reviewing control logic, and making sure hazards are recognized and managed before they become problems.
For a broader introduction to the discipline, read What Is Chemical Engineering? Definition & Scope.
This article focuses on what a chemical engineer actually does in an industrial environment, using a practical process example to show the type of reasoning behind ordinary plant equipment and daily engineering decisions.
What a Chemical Engineer Does in Practice
A chemical engineer does far more than perform calculations or size equipment. The role is defined by the ability to connect process objectives to a plant that can actually run: stable operation, acceptable performance, maintainability, controllability, and safety.
That work includes questions such as:
- how a process should operate under normal conditions
- what happens when conditions move away from design intent
- which variables must be controlled
- which equipment limitations matter in real operation
- how utilities affect performance
- how the system should respond during credible upsets
This is where chemical engineering becomes practical. A process scheme may look acceptable on paper, but an industrial plant only works when the design logic, operating envelope, and protection philosophy remain consistent.
A Practical Example: CO₂ Removal from Syngas Using Hot Potassium Carbonate
A useful way to explain what chemical engineers do is to look at a real industrial function: removing carbon dioxide from synthesis gas using a hot potassium carbonate (K₂CO₃) solvent loop, often associated with Benfield-type absorption systems.
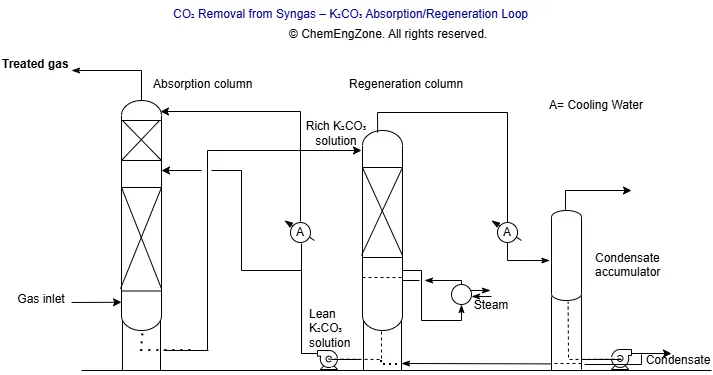
In many chemical and energy applications, synthesis gas must be conditioned before it is sent downstream to other units such as shift conversion, synthesis loops, or purification systems. One of the typical requirements is the reduction of CO₂ to a defined specification.
A hot potassium carbonate system generally includes an absorber, where CO₂ is transferred from the gas phase into the solvent, a regeneration section where the rich solution is stripped and reused, and a circulation loop with pumps and heat recovery between lean and rich streams.
This is a strong example because several layers of engineering are concentrated in the same unit: mass transfer, thermal effects, hydraulic behavior, utilities, control stability, corrosion risk, and venting philosophy. The job of the chemical engineer is not limited to choosing the main equipment. It includes defining how the whole system should perform, where its limits are, and how it remains operable under both routine and upset conditions.
For a broader overview of process equipment, see Unit Operations in Chemical Engineering: Types and Examples.
What a Chemical Engineer Checks First on This System
Before detailed design is finalized, a chemical engineer reviews the questions that determine whether the system is technically feasible and operationally robust.
CO₂ removal target and downstream constraints
The first point is the required outlet specification. How much CO₂ can remain in the treated gas before downstream units are affected? What happens if that target is not met? In addition to CO₂, the engineer may also consider water carryover, solvent traces, or contaminants that can interfere with catalysts, purification steps, or product quality.
The target is never just a number on a datasheet. It is tied to the performance and sensitivity of the next process unit.
Driving force and operating window
For carbonate-based absorption systems, performance depends strongly on temperature, pressure, and CO₂ partial pressure. The engineer must verify that the operating conditions provide a real driving force for absorption, not only a theoretical one.
A system may appear acceptable from a simplified balance, but if the actual operating window is too narrow, the unit will become unstable or inefficient whenever feed conditions drift.
Hydraulic stability and distribution inside the absorber
Column performance depends not only on chemistry, but also on hydraulic behavior. Poor liquid distribution, underestimated pressure drop, maldistribution in internals, or excessive vapor rates can all reduce separation efficiency.
An absorber that seems adequate in the calculation stage may become difficult to run if the column approaches flooding, if liquid distribution is uneven, or if the process becomes sensitive to small flow variations.
This is a typical example of what chemical engineers do: they do not stop at nominal design values, but examine whether the system will remain stable in real plant conditions.
Regeneration duty and heat integration
The regeneration section determines whether the solvent can be restored efficiently and recirculated without excessive utility consumption. Steam demand, stripping temperature, and heat recovery between lean and rich solvent streams are central design and operating questions.
This is not a secondary optimization. In many systems, heat integration determines whether the unit is economically acceptable. A chemical engineer therefore evaluates not only whether the loop works, but whether it works with a sensible energy balance.
Corrosion risk and material selection
Potassium carbonate systems can become corrosive depending on temperature, CO₂ loading, contaminants, and local operating conditions. Material selection cannot be based on average values alone. It must reflect the most severe credible conditions the system may experience.
This is where chemical engineering intersects with reliability and long-term operability. The engineer must understand the process envelope well enough to identify where corrosion is likely to accelerate and how that affects equipment life, inspection planning, and maintenance strategy.
Foaming, entrainment, and solvent losses
In absorption systems, foaming and liquid entrainment can create both process and mechanical problems. Solvent carryover may affect downstream equipment, increase operating losses, and reduce performance stability.
This is why demisters, knockout arrangements, line routing, and separator design matter. What may appear to be minor details in a process scheme can have a strong effect on reliability and housekeeping in the real plant.
Control philosophy
A plant does not run because equipment exists. It runs because the right variables are controlled in the right place. In a solvent loop, the engineer must decide how lean solvent flow is regulated, how pressure and temperature are controlled, how levels are maintained, and how the unit behaves during load changes.
Poor control philosophy often produces a plant that is technically functional but operationally unstable. A good chemical engineer therefore checks not only the process concept, but also whether the operating logic is realistic.
Relief, venting, and upset handling
Another essential question is what happens when the system moves outside normal operation. Where does CO₂ go during startup, shutdown, or upset? What happens if regeneration is interrupted, pressure rises, or utilities are lost? Are relief paths and vent destinations consistent with the protection philosophy of the plant?
This is one of the clearest examples of engineering responsibility. A system is not complete when it works during normal operation. It must also behave predictably when conditions deteriorate.
These are the questions that separate a flowsheet from a plant that can actually run.
Which Engineering Roles Are Involved

Chemical engineering in industry is rarely the work of one isolated person. Several engineering functions contribute to the final result, each with a different responsibility.
Process Engineer
The process engineer defines how the unit should operate from a technical standpoint. This includes operating conditions, process targets, thermal and hydraulic requirements, equipment sizing criteria, and checks on operability.
In the CO₂ removal system, the process engineer typically defines the required CO₂ specification, solvent circulation rate, absorber and regenerator performance targets, heat integration logic, and the operating limits needed to keep the loop stable.
For broader context on the foundations behind these decisions, see Chemical Engineering Core Disciplines – A Practical Overview.
Process Safety Engineer
The process safety engineer focuses on hazard identification, loss of containment risk, overpressure scenarios, vent handling, and the adequacy of safeguards. In a CO₂ removal loop, this can include relief routing, steam-related hazards, corrosion-driven failure scenarios, and the safe management of vented gases.
This role does not replace process engineering. It strengthens it by challenging the system under abnormal and upset conditions.
For a practical introduction to hazard review, see What Is HAZOP Analysis? Example and Template.
Project Engineer
The project engineer translates design intent into implementation. This includes coordination of piping and instrumentation works, procurement, schedule control, contractor management, interface between disciplines, and support during installation and commissioning.
Without this role, even a technically sound process design may be executed poorly in the field.
Maintenance Engineer
The maintenance engineer supports long-term reliability and availability. In solvent systems, this often means monitoring corrosion, following exchanger fouling, diagnosing repeated pump problems, and identifying failure patterns that affect plant continuity.
This role becomes especially important after startup, when the difference between a design assumption and real plant behavior becomes visible.
If you want a broader view of engineering roles across industries, read Career Opportunities in Chemical Engineering.
Conclusion
This example shows that chemical engineering is not defined by a single calculation, a simulator model, or a piece of equipment taken alone. It is defined by the ability to connect process targets to reality.
That means understanding how performance, utilities, hydraulic behavior, corrosion risk, control logic, and safety constraints interact in the same system. Industrial plants are operating systems with technical intent, physical limitations, and protection layers that must remain coherent. Chemical engineers are the professionals responsible for keeping that coherence.
⬆️ Back to TopOther Articles You May Find Useful
- What Is Chemical Engineering? Definition & Scope
- Chemical Engineering Core Disciplines – A Practical Overview
- Chemical Engineer Skills: 2 Field Practices
- Career Opportunities in Chemical Engineering
- What Is HAZOP Analysis? Example and Template
FAQ
What does a chemical engineer do in a plant?
A chemical engineer defines how a process should operate safely and effectively in real industrial conditions. This includes process targets, operating limits, equipment behavior, control strategy, and the evaluation of credible process deviations.
What does a process engineer do every day?
A process engineer typically reviews plant data, supports operations, evaluates process problems, checks equipment performance, follows process modifications, and helps ensure that the unit remains stable, efficient, and safe.
Why is operability important in chemical engineering?
Problems, checks equipment performance, follows process modifications, and helps ensure that the unit remains stable, efficient, and safe.
Add Image
Why is operability important in chemical engineering?
A process may be correct in principle but still difficult to run in practice. Operability means the plant can be controlled, maintained, and kept stable under realistic operating conditions, not only under ideal design assumptions.
What is the difference between a process engineer and a process safety engineer?
The process engineer focuses on how the unit should work. The process safety engineer focuses on how the unit can fail, what the consequences are, and whether the existing safeguards are adequate.