What is the Distillation Process?

Distillation is a method used to separate the components of a liquid mixture by exploiting differences in their volatility.
When a liquid mixture is heated, part of it vaporizes. The vapor phase is richer in the more volatile components, while the remaining liquid becomes enriched in the less volatile ones.
This behavior is governed by vapor–liquid equilibrium, which depends on the physical properties of the components and on operating conditions such as temperature and pressure.
For ideal mixtures, these equilibrium relationships can be described quantitatively using Raoult’s Law, allowing the composition of the vapor phase to be predicted from that of the liquid.
Heating provides the energy required for partial vaporization, enabling phase equilibrium to be established between liquid and vapor.
Partial Vaporization Operations (Flash)
As shown in Figure 1, a mixture composed of N components, with initial composition x01,x02,…,x0N and flow rate L0, is subjected to an initial pressure P0 and preheated to a temperature T0.
The mixture then passes through an expansion valve, where it undergoes a sudden pressure drop down to a value P.
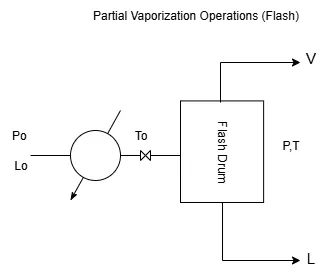
If the temperature is sufficiently high, the pressure drop results in two streams at the outlet of the flash drum:
– a liquid stream L with composition x₁, x₂, x₃, …, xn.
– and a vapor stream V with composition y₁, y₂, y₃, …, yn.
The vapor and liquid phases leaving the flash drum are in vapor–liquid equilibrium.
A flash operation therefore produces a single equilibrium separation through partial vaporization.
A distillation column extends this concept by arranging multiple equilibrium stages in series, where rising vapor and descending liquid repeatedly exchange mass and energy, progressively enhancing separation along the column height.
Distillation Column – Step-by-Step Process Illustration
How does a distillation column work? A continuous distillation column equipped with a reboiler and a condenser illustrates how separation is achieved through repeated vapor–liquid equilibrium stages, rather than a single equilibrium step as in flash vaporization.
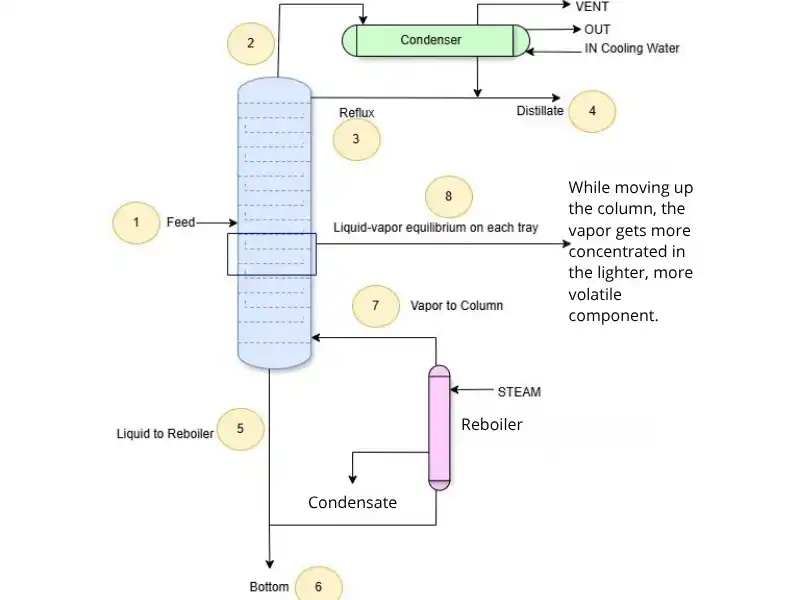
1. Feed (Entry Point)
A liquid mixture is introduced at a certain tray inside the column. This is the feed, and its position depends on the volatility of the components.
2. Vapor Rises – No Expansion Needed
Vapor generated in the reboiler rises through the column.
3. Liquid Falls
Condensed liquid from the top (reflux) and excess liquid from the feed flow downward by gravity. This creates countercurrent contact between vapor and liquid.
4. Equilibrium on Each Tray (or Packing)
At each tray (or along the packing), rising vapor and descending liquid exchange mass and energy until local vapor–liquid equilibrium is approached. Each stage, therefore, behaves as an equilibrium step driven by composition differences rather than by a pressure drop.
5. Reboiler
At the bottom, part of the liquid is vaporized by adding heat. This generated vapor re-enters the column, continuing the separation.
6. Condenser & Reflux
At the top, the vapor is condensed. Part of it is collected as distillate, and part is returned as reflux, helping maintain efficiency and sharp separation at the top trays.
In this configuration, separation results from the combined effect of heat input, phase equilibrium, and countercurrent mass transfer across multiple stages, making distillation one of the most complex unit operations in chemical engineering.
Common Types of Distillation
In industrial applications, distillation can be implemented through a wide range of configurations, depending on factors such as feed composition, thermal sensitivity, required purity, and operational constraints.
Common examples include batch and continuous systems, vacuum operation, steam-assisted configurations, and special arrangements designed to handle azeotropic mixtures.
These configurations differ in equipment layout and operating strategy, but all rely on the same fundamental principle of vapor–liquid equilibrium.
Distillation equipment
Distillation columns are designed to maintain sustained vapor–liquid contact and can be configured with different internals depending on separation requirements, operating pressure, throughput, and fluid properties.
From an engineering perspective, distillation columns are primarily classified as tray columns or packed columns, which represent the two fundamental approaches to achieving staged or continuous vapor–liquid equilibrium.
Within these families, several variants exist, including different tray designs, random or structured packing, and high-capacity internals, as well as specialized configurations such as dividing-wall, reactive, extractive, or thin-film columns developed for specific process constraints.
Regardless of the internal design, all distillation columns rely on the same underlying principle: repeated vapor–liquid equilibrium combined with countercurrent mass transfer to progressively enhance separation.
Download the PDF: Tray Columns vs Packed Columns – Engineering Comparison
A concise technical reference comparing tray columns and packed columns in terms of operating principles, efficiency, pressure drop, and typical industrial applications.
Conclusion
Distillation is often introduced as a simple separation based on differences in boiling points, but in engineering practice it is defined by vapor–liquid equilibrium, mass transfer, and process configuration.
From flash vaporization to staged columns, from tray to packed internals, the operating principle remains unchanged while the implementation adapts to process constraints and performance requirements.
Understanding how distillation works therefore means understanding not only the separation mechanism itself, but also how engineers translate that principle into different equipment layouts and operating strategies across industrial applications.
Ing. Ivet Miranda
⬆️ Back to TopOther Articles You May Find Useful
• Tray Efficiency: Improve Column Performance
• Unit Operations in Chemical Engineering: Types and Examples
• What Is Heat Transfer? Definition and Types
• The 4 Laws of Thermodynamics
FAQ
Why distillation is important?
Distillation matters because it gives us a reliable, scalable way to take a mixed liquid (or liquid + vapor) stream and pull out one or more components in a purer, usable form.
What are distillation fractions?
Distillation fractions are the different portions or cuts of a mixture that are separated during a distillation process based on their boiling points. Each fraction is collected over a specific temperature range and typically contains compounds with similar volatilities.
Distillation – How Does It Work?
Distillation works by separating the components of a liquid mixture based on differences in their boiling points. When you heat the mixture, the more volatile components vaporize first. These vapors are then condensed back into liquid and collected separately. This allows you to isolate one or more pure substances from a mixture.
Why is the distillation rate important?
The distillation rate—the speed at which vapor is condensed and collected—directly affects product quality, efficiency, and process control in both laboratory and industrial distillation. It’s not just about how fast you can collect a product, but how well the separation works.
What is the difference between tray and packed columns?
Tray columns use horizontal plates to separate the mixture step by step, while packed columns use special materials to create a continuous surface for contact. Tray columns are better for large flows and easier to clean; packed columns are more efficient for low-pressure operations and smaller systems.